Table of Contents
1. What "NMC" actually stands for
2. Reading the numbers: stoichiometry explained
3. The three-way tradeoff: capacity, stability, cost
4. NMC 532 — the conservative baseline
5. NMC 622 — the mainstream sweet spot
6. NMC 811 — high energy, high risk
7. The Jahn-Teller distortion problem in nickel-rich cathodes
8. Thermal stability: why more nickel means more danger
9. Real-world application mapping
11. References
1. What "NMC" Actually Stands For
NMC stands for Lithium Nickel Manganese Cobalt Oxide — a family of layered oxide cathode materials with the general formula LiNiₓMnᵧCo_zO₂, where x + y + z = 1.
The structure is a repeating sandwich: layers of lithium ions alternate with layers of transition metal oxide, all arranged in a rhombohedral lattice (space group R3̄m). During discharge, Li⁺ ions leave the anode, travel through the electrolyte, and intercalate back between those oxide layers in the cathode. On charge, the process reverses. The transition metals — Ni, Mn, Co — remain in the cathode layer and facilitate the electron transfer that makes the whole cycle possible.
Each metal plays a distinct electrochemical role:
Metal | Primary role | What increasing it does |
|---|---|---|
Nickel (Ni) | Capacity provider | ↑ energy density · ↓ thermal stability |
Manganese (Mn) | Structural stabiliser | ↑ cycle life and safety · ↓ capacity |
Cobalt (Co) | Electronic conductivity | Smoother kinetics · ↑ cost and supply-chain risk |
The ratio of these three metals is what the numbers in the name describe — and choosing that ratio is one of the most consequential engineering decisions in the entire battery cell design process.
2. Reading the Numbers: Stoichiometry Explained
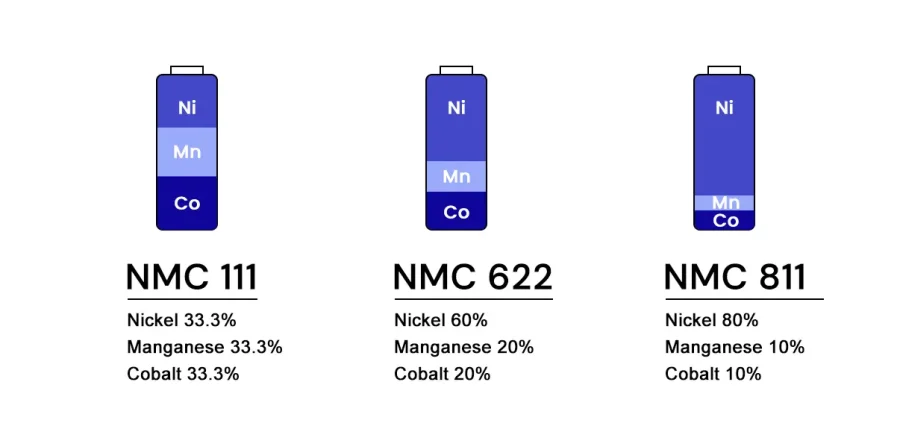
The naming convention is straightforward once you know it: NMC X-Y-Z gives the molar ratio of Ni:Mn:Co, expressed in tenths.
Variant | Formula | Ni | Mn | Co |
|---|---|---|---|---|
NMC 532 | LiNi₀.₅Mn₀.₃Co₀.₂O₂ | 50% | 30% | 20% |
NMC 622 | LiNi₀.₆Mn₀.₂Co₀.₂O₂ | 60% | 20% | 20% |
NMC 811 | LiNi₀.₈Mn₀.₁Co₀.₁O₂ | 80% | 10% | 10% |
All three share the same crystal structure. What changes is how much nickel dominates the transition metal layer. Since nickel is the primary redox-active species — it cycles between Ni²⁺, Ni³⁺, and Ni⁴⁺ as lithium is extracted and reinserted — a higher nickel fraction means more electrons transferred per formula unit, which means more capacity.
The theoretical specific capacity can be estimated from Faraday's law:
Q_theoretical = (x_Li × F) / M_formula
>
where F = 96,485 C/mol (Faraday's constant) and M ≈ 96–97 g/mol for NMC variants.
In practice, cells are not cycled to the absolute thermodynamic limits, so practical capacities fall below theoretical:
Variant | Theoretical capacity (mAh/g) | Practical capacity (mAh/g) |
|---|---|---|
NMC 532 | 155–165 | 150–160 |
NMC 622 | 170–180 | 165–175 |
NMC 811 | 200–220 | 190–200 |
3. The Three-Way Tradeoff: Capacity, Stability, Cost
The fundamental problem in NMC cathode design is that the three properties you want most — high energy density, long cycle life, and thermal safety — do not improve together. Increasing nickel content raises energy density while simultaneously reducing both cycle life and thermal stability. There is no formulation that optimises all three at once. Nickel is the dial, and every position on the dial is a compromise.
This is not a soft engineering preference. It is rooted in two physical realities that are explored in detail in sections 7 and 8: the structural phase transition that nickel-rich cathodes undergo at high states of charge, and the thermodynamic instability of the highly oxidised Ni⁴⁺ state when exposed to heat.
Property | NMC 532 | NMC 622 | NMC 811 |
|---|---|---|---|
Practical capacity (mAh/g) | 150–160 | 165–175 | 190–200 |
Cell energy density (Wh/kg) | 220–240 | 240–260 | 270–300 |
Cycle life to 80% SoH (full cycles) | 1500–2000 | 1000–1500 | 500–1000 |
Exothermic onset temperature (°C) | ~250 | ~220 | ~170–180 |
Cobalt content | 20% | 20% | 10% |
Relative raw material cost | Moderate | Moderate | Lower Co, higher Ni |
Moving from NMC 532 to NMC 811 gains roughly 25–30% more capacity per gram, halves cycle life, and drops the thermal safety margin by 80°C.
4. NMC 532 — The Conservative Baseline
NMC 532 (LiNi₀.₅Mn₀.₃Co₀.₂O₂) was the dominant commercial cathode formulation through the mid-2010s. With equal parts nickel and combined Mn+Co stabilisers, it occupies the unique position on the tradeoff curve where none of the three properties is severely compromised:
Cycle life is strong. The high manganese content stabilises the crystal structure against phase transitions during deep cycling. The 30% Mn fraction keeps Jahn-Teller-distortion-driven cracking at bay far longer than nickel-rich variants.
Thermal onset at ~250°C gives genuine headroom above realistic abuse temperatures, and makes propagation between cells far less likely in a pack-level thermal event.
Capacity is the ceiling. At ~155 mAh/g practical, achieving competitive range in an EV requires either more cells or a larger, heavier pack than NMC 622 or 811 would require.
NMC 532 is now rare in new automotive designs. You find it primarily in stationary storage systems, e-bike packs, and first-generation PHEVs where long service life mattered more than energy density.
5. NMC 622 — The Mainstream Sweet Spot
NMC 622 (LiNi₀.₆Mn₀.₂Co₀.₂O₂) is the formulation used in the majority of current-generation premium EV cells. By published cell teardown and analysis data, it includes the BMW i4, most Panasonic 2170 cylindrical cells (select variants), and LG Energy Solution's mainstream pouch products for Hyundai and Stellantis platforms.
Who uses NMC 622? BMW i4 eDrive40 · Panasonic 2170 · LG NCMA pouch (IONIQ 6) · SK On (Kia EV6 Standard Range)
The 10-percentage-point shift from Mn to Ni — relative to NMC 532 — delivers a real capacity improvement without crossing the threshold into the severe structural instability of NMC 811:
~170 mAh/g practical capacity — approximately 10% more than NMC 532 in the same cell architecture
1000–1500 full cycles to 80% SoH — adequate for automotive use, especially when BMS limits daily charging to 80% SOC (partial cycles are substantially less damaging than full 100-to-0 cycles)
~220°C exothermic onset — allows pack thermal management strategies to intercept a thermal event before it becomes self-sustaining
Cobalt content stays at 20%, the same as NMC 532. There is no cost change from switching between these two; the gain is entirely from replacing less-active Mn with capacity-providing Ni.
6. NMC 811 — High Energy, High Risk
NMC 811 (LiNi₀.₈Mn₀.₁Co₀.₁O₂) is the aggressive end of the current commercial NMC spectrum. 80% nickel content delivers cell-level energy densities of 270–300 Wh/kg — roughly 15–25% more than NMC 622 in the same cell format. That is the reason it is used in applications where maximum range per kilogram is non-negotiable.
Who uses NMC 811? LG M50/M50LT in Tesla Model 3 Long Range (21700 cylindrical) · SK Innovation in Kia EV6 Long Range · CATL NMC811 in various Chinese-market EVs · Samsung SDI in BMW iX M60
The performance premium comes at a real cost. Capacity fade follows an exponential decay model:
Q(n) = Q₀ · e^(−αn)
>
where Q(n) is capacity at cycle n, Q₀ is initial capacity, and α is the degradation rate constant.
>
For NMC 811 at full cycles, 25°C: α ≈ 0.0008–0.0015 per cycle
For NMC 622 at full cycles, 25°C: α ≈ 0.0004–0.0007 per cycle
At α = 0.0012 (midpoint NMC 811), the cell reaches 80% of initial capacity at approximately n = −ln(0.8) / 0.0012 ≈ 186 full cycles. In practice, BMS charge limits and partial-cycle usage extend this significantly — but the relative disadvantage versus NMC 622 is real and measurable.
Reduced cobalt also cuts electronic conductivity across the cathode particle, requiring careful particle engineering to compensate: single-crystal morphology, LiNbO₃ or Al₂O₃ surface coatings, and tighter sintering controls. These are achievable but add process complexity and cost that partially offset the raw material savings from less cobalt.
7. The Jahn-Teller Distortion Problem in Nickel-Rich Cathodes
This is the fundamental structural mechanism behind NMC 811's faster capacity fade, and it deserves a precise explanation.
During charging, lithium ions are extracted from the NMC cathode layer. As Li⁺ leaves, the transition metals oxidise to maintain charge neutrality. In nickel-rich NMC, this means nickel progresses through:
Ni²⁺ → Ni³⁺ → Ni⁴⁺ as lithium extraction increases
At high states of charge — above approximately 4.2 V vs Li/Li⁺ for NMC 811 — the accumulation of Ni⁴⁺ in the lattice forces a structural phase transition from the hexagonal H2 phase to the hexagonal H3 phase. This transition is accompanied by a ~5% contraction of the c-axis lattice parameter — the dimension perpendicular to the lithium ion planes.
Five percent sounds small. At the particle level, it is mechanically catastrophic when repeated.
Each charge-discharge cycle takes the crystal through this expansion-contraction sequence. After hundreds of cycles, the cumulative volumetric fatigue generates microcracks that propagate through the polycrystalline secondary cathode particles — the ~10–15 μm agglomerates of smaller primary crystallites that make up the active material. The consequences compound rapidly:
1. Fresh NMC surface is exposed to electrolyte — a new cathode-electrolyte interphase (CEI) layer forms, irreversibly consuming lithium from the active inventory
2. Cracked fragments are electrically isolated — any material disconnected from the conductive network stops contributing to capacity permanently
3. Each crack seeds the next — stress concentrations at crack tips initiate further fracture at lower mechanical loads in subsequent cycles
The result is non-linear capacity fade — not the gentle, predictable drift seen in NMC 622, but an increasingly steep drop once crack networks become pervasive. This is the origin of the "knee point" visible in NMC 811 capacity fade curves at ~500–800 full cycles under aggressive cycling conditions.
Single-crystal NMC 811 (SC-NMC 811) is the direct engineering response. By replacing polycrystalline secondary particles with large single-crystal primaries — typically 2–5 μm diameter — the internal grain boundaries where cracks initiate are eliminated entirely. SC-NMC 811 cycle life approaches NMC 622 performance in controlled test conditions. The barriers to mainstream adoption remain manufacturing yield (single-crystal synthesis is harder to control than coprecipitation) and cost.
8. Thermal Stability: Why More Nickel Means More Danger
The exothermic decomposition of a fully charged NMC cathode is the triggering event in thermal runaway. As the cathode heats above a chemistry-specific onset temperature, it releases oxygen and heat through:
LiₓNiO₂ → NiO (rock-salt structure) + ½O₂ + heat
The released oxygen then reacts exothermically with the organic electrolyte solvent — typically ethylene carbonate / dimethyl carbonate (EC/DMC) mixtures:
EC + O₂ → CO₂ + H₂O + ΔH (strongly exothermic)
This two-step sequence is self-sustaining above the onset temperature, which is why it matters so much for pack-level safety. The onset temperatures by variant:
Chemistry | Exothermic onset (°C) | Margin above 100°C pack temp |
|---|---|---|
LFP | ~270 | 170°C |
NMC 532 | ~250 | 150°C |
NMC 622 | ~220 | 120°C |
NMC 811 | ~170–180 | 70–80°C |
NMC 811's onset is only 70–80°C above the peak temperatures a pack can reach during aggressive DC fast charging. That narrow margin means a locally overheated cell — from a manufacturing defect, a damaged separator, or a cooling failure — can generate enough heat to push adjacent cells above their own onset temperature. A cascading thermal runaway across multiple cells is the result.
LFP's 270°C onset is the core reason it is considered inherently safer for large-format packs: the same local heating event is far less likely to propagate.
Pack designers using NMC 811 must choose one of three responses to this narrowed margin: add more inter-cell thermal isolation mass (which reduces pack-level energy density), deploy faster active cooling capable of intercepting temperature rise before the 170°C threshold, or accept higher propagation risk and design the pack enclosure for containment rather than prevention.
9. Real-World Application Mapping
The commercial deployment of NMC variants reflects the tradeoff directly: where cost-per-kWh matters most, NMC 532 or LFP wins. Where range-per-kilogram dominates, NMC 811 is the answer. NMC 622 holds the middle ground where neither extreme is acceptable.
Application | Typical variant | Primary selection reason |
|---|---|---|
Grid-scale stationary storage | NMC 532 or LFP | Cycle life and thermal safety over density |
E-bikes and scooters | NMC 532 | Cost, abuse tolerance, moderate cycle count |
PHEV (small pack) | NMC 622 | Density matters for the small pack; moderate cycle requirement |
BEV mainstream | NMC 622 | Balanced range, cycle life, and safety |
BEV long-range / performance | NMC 811 | Maximum range per kg is the dominant constraint |
BEV cylindrical cells (e.g. Tesla LR) | NMC 811 (LG M50LT) | Cylindrical format dissipates heat more evenly, managing NMC 811's thermal risk better than large-format pouch |
10. What Comes After 811?
The commercial trajectory points clearly toward NMCA (nickel-manganese-cobalt-aluminium) and near-zero-cobalt NMx formulations that push nickel content above 90%. Aluminium doping and advanced surface coatings — LiNbO₃, Al₂O₃, lithium-reactive fluoride layers — are used to stabilise the surface against electrolyte attack and slow the H2→H3 phase transition kinetics, compensating partially for the reduced manganese and cobalt content.
Panasonic's 4680 cells for Tesla incorporate a high-nickel chemistry optimised around the cylindrical format's superior radial heat dissipation. The large-format cylindrical geometry allows NMC 811-class chemistry to be used more aggressively than it could be in a pouch cell of comparable energy.
CATL's Qilin battery and BYD's Blade battery go in a different direction entirely — rather than pushing cell-level chemistry, they maximise LFP energy density at the pack level through structural cell-to-pack (CTP) integration that eliminates modules and uses the cells themselves as structural members. At the pack level, the energy density gap between LFP and NMC 811 narrows significantly: LFP CTP packs achieve 150–160 Wh/kg at pack level, versus 180–200 Wh/kg for NMC 811 module-based packs.
That competition — structural LFP at the pack level versus high-nickel NMC at the cell level — is the defining tension in high-energy EV battery design heading into the second half of the decade.
The unanswered question remains: does single-crystal NMC 811 represent the practical ceiling for layered oxide cathodes, or can further nickel enrichment combined with advanced surface engineering genuinely shift the tradeoff curve rather than merely moving along it?
References
1. Noh, H-J. et al. — "Comparison of the structural and electrochemical properties of layered Li[NixCoyMnz]O2 cathode material for lithium-ion batteries," Journal of Power Sources, 233, 2013. https://doi.org/10.1016/j.jpowsour.2013.01.063
2. Li, W. et al. — "The volcano effect of Ni content in NMC cathodes," Advanced Energy Materials, 2020. https://doi.org/10.1002/aenm.202000034
3. Ryu, H-H. et al. — "Capacity fading of Ni-rich Li[NixCoyMn1-x-y]O2 cathodes: effect of Li/Ni disorder," Chemistry of Materials, 2018. https://doi.org/10.1021/acs.chemmater.8b00812
4. Märker, K. et al. — "Evolution of structure and lithium dynamics in LiNi0.8Mn0.1Co0.1O2 during electrochemical cycling," Chemistry of Materials, 2019. https://doi.org/10.1021/acs.chemmater.9b01971
5. Battery University — "BU-205: Types of Lithium-ion." https://batteryuniversity.com/article/bu-205-types-of-lithium-ion
6. Manthiram, A. et al. — "Nickel-rich and lithium-rich layered oxide cathodes: progress and perspectives," Advanced Energy Materials, 2016. https://doi.org/10.1002/aenm.201501010
7. Liu, T. et al. — "Origin of structural degradation in Li-rich layered oxide cathode," Nature, 606, 2022. https://doi.org/10.1038/s41586-022-04689-y 8. Blomgren, G.E. — "The Development and Future of Lithium Ion Batteries," Journal of The Electrochemical Society, 164(1), 2017. https://doi.org/10.1149/2.0251701jes